
RED LIGHT DONE RIGHT
WHERE TECHNOLOGY MEETS WELLNESS
Promote your products


Generate Additional Revenue
-
Boost profits with a high-demand service that’s easy to integrate.
-
Red light therapy requires minimal training and has low operating costs.

Increase Client Retention
-
Keep clients loyal with innovative services.
-
Add variety to offerings to encourage frequent visits.

Enhance Client Experience
-
Help your clients temporarily ease their muscle pain and increase blood flow with a rejuvenating experience.
-
Stand out with advanced technology that sets you apart from your competitors
Click Here To Find Out How

FDA Registered
At Body Balance System, we're committed to providing red light therapy systems that meet the highest safety and quality standards. Our systems are FDA registered, reflecting our dedication to compliance with federal regulations and demonstrating our commitment to excellence. This registration provides your wellness business with the confidence that you are offering clients a trusted and reliable treatment option.
FDA Registration Number #3010627475
NRTL Certified
Beyond FDA registration, we go the extra mile to ensure the safety and performance of our products. Our system, the OvationULT bed, undergoes rigorous testing by Nationally Recognized Testing Laboratories (NRTLs) like SGS – a global leader in product safety certification. These independent labs evaluate our systems for EMF, radiation, electrical safety, and FCC compliance. This meticulous third-party verification guarantees our reported outputs are accurate and validated, giving you and your clients peace of mind and the most effective red light therapy experience possible.
NRTL Certification Number: SGSNA/25/SUW 00264
Proudly Manufactured in the USA. Prices unaffected by tariffs.
Financing Options to Grow Your Business
Financing options are subject to approval and available for those who qualify.
Discover the Power of Red Light Therapy
Learn how red light therapy can enhance your wellness practice and elevate client experiences.
What Our Clients Are Saying
-
 Debbie J.
Debbie J.I've used Body Balance System for 5 years. One of my clients with a recurring brain tumor saw the spot disappear after regular foot baths—nothing else changed. The doctors were amazed!
-
 Shari B.
Shari B.After Rocky Mountain Spotted Fever, my joint pain vanished after a few sessions. Sinus issues are gone too. Perfect for professional use—reliable, easy to clean, and great service.
-
 Scott K.
Scott K.Chronic knee pain made work impossible until I tried Body Balance System. One foot bath eased my pain, letting me move again. Highly recommend!
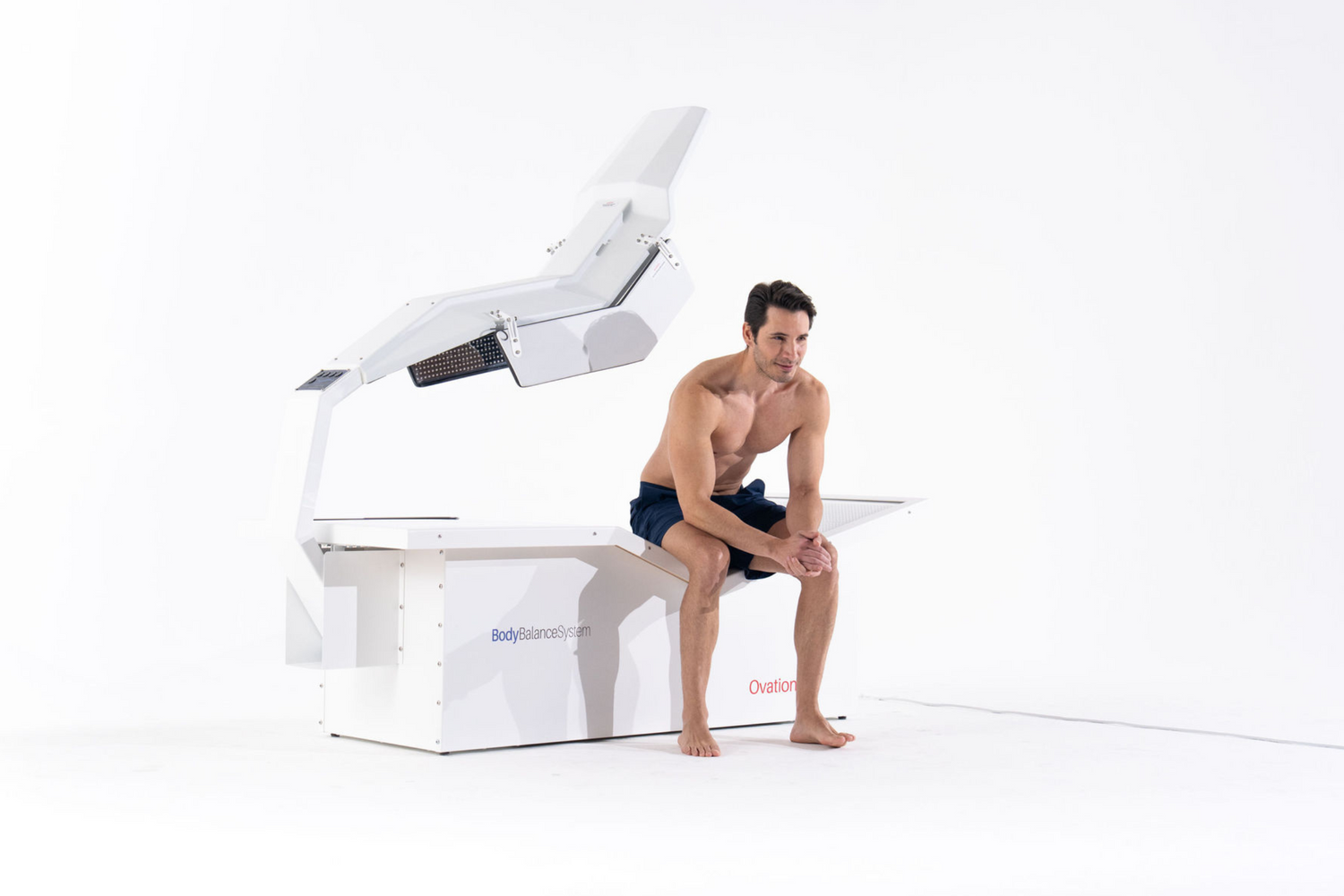
Why Choose Us
At Body Balance System, we are dedicated to empowering wellness practices with innovative solutions. Our commitment to quality, performance, and customer satisfaction sets us apart in the industry, ensuring that you receive the best products and support for your business.
Our Priority
FDA Registered Products
Our systems meet rigorous safety and efficacy standards, giving you peace of mind when offering treatments to your clients.
Handcrafted in the USA
Each of our products is meticulously crafted in Las Vegas, ensuring quality and attention to detail that mass-produced alternatives can't match.
Innovative Technology
We continuously invest in research and development to provide cutting-edge solutions that enhance the wellness experience for your clients.
Exceptional Customer Support
Our dedicated team is here to support you every step of the way, from product selection to training and beyond.

Satisfied Clients
Years in Business
Wellness Partners
Innovative Products
Innovative Wellness Solutions for Your Business
At Body Balance System, we specialize in advanced solutions designed to elevate wellness experiences and deliver exceptional results.
-
 Red Light TherapyOur advanced red light therapy systems use state-of-the-art technology to provide non-invasive treatments that enhance wellness. With high-quality diodes for optimal performance and ergonomic designs for client comfort, these systems ensure maximum light penetration and a relaxing experience.
Red Light TherapyOur advanced red light therapy systems use state-of-the-art technology to provide non-invasive treatments that enhance wellness. With high-quality diodes for optimal performance and ergonomic designs for client comfort, these systems ensure maximum light penetration and a relaxing experience. -
 Detox SolutionsOur detox solutions offer a holistic approach to cleansing and rejuvenating the body. These solutions are designed to help individuals eliminate toxins and achieve optimal levels of balance and health. Experience the benefits of detoxification and take a proactive step towards enhancing your wellness journey.
Detox SolutionsOur detox solutions offer a holistic approach to cleansing and rejuvenating the body. These solutions are designed to help individuals eliminate toxins and achieve optimal levels of balance and health. Experience the benefits of detoxification and take a proactive step towards enhancing your wellness journey.
Featured Press

Red Light Therapy Locator
Explore our network of wellness practices that offer our advanced red light therapy products. Find a location near you where to experience our innovative solutions.

Which Red Light Therapy Product is Right for Your Practice?
Take our quick quiz to discover the solution that aligns with your wellness practice goals. Our tailored recommendations will help you choose the right product to elevate your services and support your clients' well-being.
Stay Informed with Our Blog
-
 June, 2026Wavelength Guide: What 635nm and 850nm Actually Do (And Why It Matters for Commercial Use)
June, 2026Wavelength Guide: What 635nm and 850nm Actually Do (And Why It Matters for Commercial Use)Photobiomodulation (PBM) research has identified wavelength as the single most consequential variable in light-based therapy protocols. Published literature indicates that 635nm red light and 850nm near-infrared (NIR) light interact with tissue through different absorption pathways and reach different anatomical depths. For commercial operators running full-body sessions, understanding these differences is not just academic. It directly informs session design, client outcomes, and how to evaluate equipment specifications. This article translates the peer-reviewed evidence on both wavelengths into practical knowledge for spa directors, wellness operators, and fitness facility managers evaluating full-body red light systems.
What Does 635nm Red Light Actually Do in the Body?
The 635nm wavelength sits near the peak of the visible red spectrum and falls within what researchers call the "optical window" of biological tissue. According to a widely cited review by Hamblin and de Freitas published in the IEEE Journal of Selected Topics in Quantum Electronics, cytochrome c oxidase (CCO), the terminal enzyme in the mitochondrial electron transport chain, absorbs light across red and near-infrared wavelengths. It shows particular photosensitivity in the red band around 620 to 680nm. The leading hypothesis is that photon absorption dissociates inhibitory nitric oxide from CCO, restoring electron transport and increasing adenosine triphosphate (ATP) production at the cellular level.
Tissue penetration at 635nm is generally reported in the literature as approximately 1 to 2mm into skin and superficial tissue layers. This makes 635nm highly relevant for applications where the physiological targets are close to the surface, such as dermal fibroblasts, keratinocytes, superficial capillary beds, and epidermal structures. A 2024 comprehensive review published in the International Journal of Molecular Sciences found that PBM in the red band stimulates cellular chromophores involved in local circulation pathways.
For commercial operators, the practical implication is that 635nm red light addresses the outermost physiological targets in a full-body session. Published evidence positions this wavelength as well-suited for superficial muscle tissue and skin applications where shallow penetration is an asset. The depth profile is predictable and well-characterized in peer-reviewed literature, making dosimetry more manageable at this wavelength than at longer ones.
What Does 850nm Near-Infrared Light Do Differently?
The 850nm wavelength falls in the near-infrared band, outside visible human perception, and behaves fundamentally differently in biological tissue than red light. Published research on tissue optics indicates that longer wavelengths scatter less and penetrate significantly deeper. Reported penetration depths for 850nm NIR range from approximately 3 to 5cm in soft tissue under standardized conditions, depending on tissue composition, hydration, and the anatomical site. A review published in Photobiomodulation, Photomedicine, and Laser Surgery examining penetration profiles across multiple wavelengths confirmed that NIR wavelengths consistently outperform visible red light for reaching deeper anatomical structures.
At 850nm, the primary absorption target remains cytochrome c oxidase, but the geometry of that interaction changes because the light reaches tissue layers that 635nm cannot. Hamblin's 2018 review in Photochemistry and Photobiology described how PBM using near-infrared wavelengths acts on mitochondria in deeper musculoskeletal tissue, with implications for joint capsules, tendons, and deeper muscle groups that are inaccessible to red light. The biphasic nature of cellular response is especially important at 850nm. Beneficial effects depend on reaching an adequate but not excessive dose at the target tissue, and overdosing at the surface risks underperforming at depth.
For a commercial full-body system, 850nm NIR expands the physiological reach of a session beyond the skin surface. Published protocols in sports medicine and physical therapy research increasingly use NIR wavelengths when targeting deeper musculoskeletal structures, and the broader clinical literature consistently distinguishes the depth-profile advantages of NIR over visible red light for these applications. Operators evaluating systems for clientele with musculoskeletal or joint-related goals should understand that 850nm coverage is not interchangeable with 635nm coverage.
Why Does Penetration Depth Matter for Commercial Full-Body Sessions?
Penetration depth determines which anatomical targets a session can plausibly reach, and that determines the physiological applications a device can support. A system emitting only 635nm can address superficial targets with precision but cannot reach the joint capsules, synovial tissue, or deeper muscle bellies that lie beyond a few millimeters of tissue depth. Conversely, a system emitting only 850nm may overdose superficial structures while trying to deliver an appropriate dose at depth, creating an uneven irradiance profile across the client's full tissue spectrum.
Published dosimetry literature frames the challenge as matching wavelength to target depth. The concept of the therapeutic window for each wavelength is well-defined in peer-reviewed research: energy delivered above it inhibits cellular response, while energy below it fails to elicit one. For a commercial operator running a two-client-per-hour throughput model, wavelength selection is the primary lever for determining which tissue layers receive a therapeutic-range dose within a fixed session window.
The dual-wavelength approach, combining 635nm and 850nm in a single full-body session, has become standard in commercial PBM protocols because it addresses both the superficial and deeper target layers simultaneously. Published research treats the two wavelengths as complementary rather than interchangeable, each contributing to a different stratum of the overall dose delivered during a session. Operators should treat dual-wavelength coverage as a fundamental baseline specification.
What Is the Biphasic Dose-Response Curve?
The biphasic dose-response curve, also called the Arndt-Schulz curve in PBM literature, describes a fundamental property of photobiomodulation: at low doses, cellular stimulation increases, but at higher doses, the same tissue can become inhibited or return to baseline. This is not unique to light therapy. It is a well-characterized phenomenon in pharmacology and biophysics. In PBM, a 2009 study by Huang, Chen, Carroll, and Hamblin published in Dose-Response first formally characterized the biphasic pattern, and subsequent updates confirmed it across multiple tissue types and irradiance levels.
The practical consequence for commercial operators is that higher irradiance and longer sessions are not automatically better. The evidence suggests that each tissue type has an optimal dose range measured in Joules per square centimeter (J/cm²), and exceeding it produces diminishing or counterproductive results. Session parameters are not interchangeable. Irradiance and duration must be specified together, and adjusting one without the other risks falling outside the effective dose range for the target tissue.
Published research uses the formula:
Dose (J/cm²) = [Irradiance (mW/cm²) x Time (seconds)] / 1000
This means that irradiance specification is inseparable from time specification in any credible commercial PBM protocol. Equipment with higher irradiance can deliver the same Joule-per-square-centimeter dose in a shorter session, which directly improves operator throughput. The biphasic curve also explains why the literature emphasizes precision, as the difference between a stimulatory and an inhibitory dose can be smaller than operators expect.
How Does Irradiance Specification Connect to Commercial Throughput?
Irradiance, measured in milliwatts per square centimeter (mW/cm²), is the rate at which light energy is delivered to the tissue surface. It is distinct from total dose (J/cm²), which also incorporates time. In commercial PBM operations, irradiance is the specification that determines how quickly a therapeutic-range dose can be accumulated during a fixed-length session window. A device with low irradiance requires proportionally longer sessions to accumulate an equivalent dose, whereas a device with higher irradiance achieves the same dose in less time.
The OvationULT operates at 65 mW/cm² irradiance. At this specification, a client can accumulate a meaningful dose within 10 to 20 minutes, which is the session window consistent with published commercial protocols for full-body PBM. For an operator running a standard two-client-per-hour model, this throughput is achievable without compromising dose delivery. If irradiance were substantially lower, either session times would need to extend beyond commercially viable windows, or clients would receive a sub-therapeutic dose per the published dose-response literature.
For operators evaluating competing systems, the irradiance specification should always be read alongside the recommended session length. A device claiming short session times while listing low irradiance presents parameters that do not align with the published dose literature. A device with 65 mW/cm² irradiance and a 10 to 20 minute session window presents a dosimetric profile consistent with the full-body photobiomodulation evidence base.
Why Do Commercial Protocols Use Both 635nm and 850nm Together?
The rationale for combining 635nm and 850nm in commercial full-body systems is comprehensive depth coverage. The two wavelengths together address the full anatomical range from the skin surface to deep musculoskeletal tissue. Published research does not position one wavelength as superior to the other in an absolute sense. Rather, the literature establishes that each is optimal for a different depth range, and a full-body system that covers only one range leaves vital physiological targets unaddressed during the session.
A 2014 paper by Karu noted that multiple wavelengths act on both mitochondrial and non-mitochondrial photoacceptors, and that tissue heterogeneity across the body means no single wavelength uniformly addresses all targets in a full-body session. Dual-wavelength coverage is therefore a structural feature of a proper protocol, not an optional enhancement. Clients with skin-focused goals draw primarily from 635nm coverage, while clients with musculoskeletal goals draw from 850nm. Most clients benefit from both simultaneously.
The OvationULT emits both 635nm red and 850nm near-infrared in a single session, delivering the dual-wavelength coverage that the published literature identifies as standard for commercial full-body photobiomodulation. The device carries FDA Registration #3010627475 (ILY product code) as a Class II medical device, reflecting strict regulatory oversight of the manufacturing and quality processes. Registered indications for the OvationULT include topical heating, temporary relief of minor muscle and joint pain and stiffness, temporary relief of minor arthritis pain, relaxation of muscle spasms, and temporary increase of local circulation.
Wavelength Comparison: 635nm vs 850nm at a Glance
Feature
635nm (Red)
850nm (Near-Infrared)
Tissue penetration (reported)
~1 to 2mm
~3 to 5cm
Primary chromophore
Cytochrome c oxidase (red band)
Cytochrome c oxidase (NIR band)
Primary tissue targets
Skin, epidermis, superficial capillaries
Deep muscle, joints, tendons
Visibility
Visible red
Invisible to the naked eye
Dose consideration
Lower scatter; predictable surface dose
Greater scatter correction needed for depth dosimetry
Commercial role
Superficial target coverage
Deep-tissue target coverage
FAQ: 635nm and 850nm in Commercial Red Light TherapyWhat is the difference between 635nm and 850nm light in PBM research?
Published research indicates that 635nm red light penetrates approximately 1 to 2mm into tissue and acts primarily on superficial structures, including skin and epidermal layers. Meanwhile, 850nm near-infrared light penetrates approximately 3 to 5cm, reaching deeper musculoskeletal tissue, joints, and tendons. Both wavelengths target cytochrome c oxidase in the mitochondrial respiratory chain, but their anatomical reach differs significantly.
Why does the wavelength specification matter when choosing a red light therapy system?
Wavelength determines where in the body light energy is absorbed. A system emitting only one wavelength addresses only one depth range. A system emitting both 635nm and 850nm addresses the full depth spectrum from the skin surface to deep tissue in a single session. For operators running a general clientele with varied goals, dual-wavelength coverage means each session delivers energy across the complete physiological target range.
What does "biphasic dose-response" mean in practice for an operator?
The biphasic dose-response curve means that cellular stimulation from PBM increases with dose up to an optimal point, then declines or reverses at higher doses. For operators, this means that longer sessions at high irradiance are not categorically better than properly calibrated shorter sessions. Dose (J/cm²) is the product of both irradiance and time, so they must be specified together. The OvationULT's 65 mW/cm² irradiance paired with 10 to 20 minute sessions is designed to deliver a dose within the published stimulatory range without overshooting into inhibitory territory.
Is the OvationULT FDA registered for photobiomodulation?
The OvationULT is FDA registered (Registration #3010627475), which reflects regulatory oversight of the device as a Class II medical device. The device's registered indications include topical heating, temporary relief of minor muscle and joint pain and stiffness, temporary relief of minor arthritis pain, relaxation of muscle spasms, and temporary increase of local circulation.
How does 65 mW/cm² irradiance affect session length and operator throughput?
At 65 mW/cm², the OvationULT accumulates dose at a rate sufficient to reach the published therapeutic range within a 10 to 20 minute session. This means a standard two-client-per-hour throughput model is entirely achievable. Systems with lower irradiance require extended sessions to reach an equivalent dose, which reduces throughput, increases per-client room time, and creates scheduling constraints that hurt commercial profitability.
Sources
-
Hamblin MR, de Freitas LF. "Proposed Mechanisms of Photobiomodulation or Low-Level Light Therapy." IEEE Journal of Selected Topics in Quantum Electronics. 2016. PMC5215870. https://pmc.ncbi.nlm.nih.gov/articles/PMC5215870/
-
Hamblin MR. "Mechanisms and Mitochondrial Redox Signaling in Photobiomodulation." Photochemistry and Photobiology. 2018. PMC5844808. https://pmc.ncbi.nlm.nih.gov/articles/PMC5844808/
-
Huang YY, Chen AC, Carroll JD, Hamblin MR. "Biphasic Dose Response in Low Level Light Therapy." Dose-Response. 2009. PMC2790317. https://pmc.ncbi.nlm.nih.gov/articles/PMC2790317/
-
Karu TI. "Cellular and Molecular Mechanisms of Photobiomodulation (Low-Power Laser Therapy)." IEEE Journal of Selected Topics in Quantum Electronics. 2014. DOI: 10.1109/JSTQE.2013.2273411. https://ieeexplore.ieee.org/document/6603355/
-
Naharro-Rodriguez J, et al. "Unlocking the Power of Light on the Skin: A Comprehensive Review on Photobiomodulation." International Journal of Molecular Sciences. 2024. PMC11049838. https://pmc.ncbi.nlm.nih.gov/articles/PMC11049838/
Related Resources
-
-
 June, 2026Red Light Therapy for Recovery Centers: Throughput, Client Experience, and ROI
June, 2026Red Light Therapy for Recovery Centers: Throughput, Client Experience, and ROIRecovery centers built around athletic performance and longevity already serve clients who want red light therapy (RLT). The modality fits your existing stack without adding operational friction. Sessions run 10 to 20 minutes, the device plugs into a standard 120V outlet, and staff training is minimal. For operators running membership models between $150 and $300 per month, RLT can be included at no extra cost to anchor retention, or offered as a premium add-on. This guide walks through the throughput math, membership integration, ROI timeline, and the questions every recovery center operator should ask before signing a purchase order.
Why Are Recovery Centers Such a Natural Fit for Red Light Therapy?
Recovery centers have built their business model around stacking modalities that help clients return to baseline after hard training. Cold plunge, infrared sauna, compression boots, cryotherapy, IV therapy, and hyperbaric chambers all share the same goal: reduce the cost of physical effort and help clients come back sooner.
Red light therapy fits that logic without friction. The device is passive, requires no practitioner contact, and delivers a repeatable client experience every session. According to data from the Global Wellness Institute, the global wellness economy represents one of the fastest-growing segments in fitness and lifestyle.
Chains such as Restore Hyper Wellness, Perspire, Remedy Place, and Sweat House LA have demonstrated that multi-modality recovery is a scalable membership business, not a boutique experiment. When a client's gym already offers compression and percussion massage, your center needs a concrete reason to hold their membership. RLT delivers that reason without requiring a licensed practitioner, a consumable supply chain, or a massive footprint.
From a compliance standpoint, commercial RLT devices must meet federal photonic safety standards. The OvationULT carries FDA Registration #3010627475 and is classified as a Class II device with an ILY product code. Body Balance System is FDA registered, which is the correct regulatory designation for photonic medical devices of this class. Confirm equivalent credentials from any vendor before buying.
How Does Red Light Therapy Fit Into an Existing Recovery Modality Stack?
Sequencing matters, and RLT has a clear position in most recovery protocols. The most common sequence starts with training, transitions to red light for local circulation and muscle relaxation, and then moves into contrast therapy like a cold plunge or cryotherapy. Research published in PubMed-indexed journals on photobiomodulation (PBM) has examined local circulation, muscle relaxation, and tissue response. Operators should attribute those findings to general research rather than to a specific device.
Within the ILY scope for the OvationULT, the device is indicated for:
-
Topical heating
-
Temporary relief of minor muscle and joint pain and stiffness
-
Temporary relief of minor arthritis pain
-
Relaxation of muscle spasms
-
Temporary increase of local circulation
Those indications map directly onto what a post-workout athlete wants: a comfortable 10 to 20 minute session that delivers warmth and relaxation before the cold plunge or the drive home.
Infrared sauna is a natural complement rather than a competitor. Some operators offer RLT first because of its shorter session and lower thermal load, followed by the infrared sauna, and finally a cold plunge as the final contrast. Others pair RLT and compression sleeves in adjacent rooms on overlapping time slots to maximize throughput per square foot. Because the OvationULT runs 10 to 20 minute sessions, the timing aligns neatly with most sauna and compression durations. This ensures clients move through the rotation without awkward waits.
What Does the Throughput Math Actually Look Like?
At a standard 15-minute session within the 10 to 20 minute range, one OvationULT supports 2 clients per hour. Over a 10-hour operating day, that is 20 client sessions per unit. A 12-hour operating day yields 24 sessions per unit. That is the baseline capacity number you need before running a meaningful ROI calculation.
The revenue side depends on how you package RLT. Recovery centers typically choose one of three models:
-
Include RLT in all membership tiers to anchor retention.
-
Offer it as a premium tier upgrade at $20 to $40 per month.
-
Price individual sessions at $25 to $45 for non-members and day-pass clients.
At $35 per session and 60 percent utilization on a 12-hour day, a single unit generates roughly $300 per day, or approximately $9,000 per month. With the OvationULT priced at $59,997, that puts the gross revenue payback period well within the 9 to 18 month range. This is before you even account for membership retention lift, which is often the larger driver.
Staff involvement per session is minimal. The client activates the session, the operator sets the protocol, and the device runs without supervision. That means you are not staffing to RLT capacity the way you would for IV therapy or manual massage. The labor efficiency is one reason RLT has a better margin profile than most other modalities at comparable price points. For a center already paying for front-desk staff to manage cold plunge rotations, adding RLT does not require a headcount increase.
What Membership Pricing Models Work Best for Recovery Centers Adding RLT?
Membership pricing in the recovery center vertical has settled around a few predictable structures:
-
Unlimited Tiers: Run $200 to $300 per month with full modality access.
-
Mid-Tier Memberships: Sit at $150 to $200 per month, covering core modalities like cold plunge, infrared sauna, and compression, but gating premium add-ons like RLT behind an upgrade.
-
Entry Tiers: Run $100 to $150 per month and are typically visit-based, functioning more like discounted punch cards.
The most common strategy is to include RLT in the top one or two membership tiers and market it as the primary differentiator. This lifts average revenue per member as existing members upgrade, and it gives prospects a concrete reason to choose the higher tier. According to ISPA industry data, service bundling is one of the highest-leverage retention tools in facility-based wellness. RLT suits bundling perfectly because it carries no meaningful consumable costs after the hardware purchase.
For day-pass and drop-in clients, individual session pricing at $30 to $45 positions RLT above compression in perceived value but below cryotherapy or IV therapy in intensity. That price point works because the session delivers a tangible experience of warmth and deep relaxation that clients love to talk about. A client who completed a cold plunge and a red light session has a specific story to tell, and that specificity drives referrals.
What Are the Space and Power Requirements for a Commercial RLT Unit?
The OvationULT is a full-body panel system designed for commercial use with a footprint that fits in a standard treatment room or wellness bay. The device runs on a standard 120V outlet. This means no electrical infrastructure upgrades, no dedicated circuit negotiations with your landlord, and no construction permitting delays.
For operators in leased spaces, this is a massive deployment advantage. Adding cryotherapy, hyperbaric oxygen, or high-amperage infrared saunas often requires electrical panel upgrades that add thousands in cost and months of delays to a buildout.
Most operators allocate a private room or semi-private bay for RLT to support the relaxation component of the session. Clients using RLT after a cold plunge often want a quiet, warm environment to complete the contrast cycle, and a shared open-floor configuration works against that experience. An 80 by 31 inch footprint fits comfortably in a standard treatment room.
Protocol simplicity is a real operational advantage. Sessions are 10 to 20 minutes, the device settings are operator-configured in advance, and clients follow a simple orientation script. There is no hands-on contact, no injectable, and no recovery observation period. The net result is that one front-desk staff member can manage RLT rotations alongside other check-in duties without dedicated practitioner time.
What Does the ROI Calculation Look Like?
The ROI framework for RLT in a recovery center has three inputs: hardware cost, revenue per unit per month, and membership retention lift. The OvationULT is priced at $59,997 with a 5-year warranty, so operators can amortize the cost over 60 months with near-zero maintenance expense. Amortized over 60 months, the monthly capital cost is approximately $1,000.
Scenario
Daily Sessions
Utilization
Session Rate
Monthly Revenue
Monthly Capital Cost
Net Monthly
Conservative (membership incl.)
12
50%
$0 (included)
Retention value est. $3K+
$1,000
Retention-driven
Moderate (mix of members + drop-in)
16
67%
$20 avg. blended
$9,600
$1,000
$8,600
Active (premium tier + drop-in)
20
83%
$30 avg. blended
$18,000
$1,000
$17,000
Retention value is often the larger number. If RLT in the top tier keeps 10 percent of a 300-member base for just one additional month per year at $250 per month, that is $7,500 in annualized retained revenue. Combined with direct session revenue, payback on a premium commercial unit falls well inside 12 to 18 months at moderate utilization.Body Balance System installations at world-class luxury resorts and spas reflect the commercial durability expectations appropriate for a device at this price point. Operators should apply the same due diligence to any purchase: confirm the FDA registration number, ask for commercial references, and get warranty terms in writing before signing.
What Should Recovery Center Operators Ask Any RLT Manufacturer Before Buying?
Buyer due diligence on commercial RLT hardware is not complicated, but most operators skip it.
-
Regulatory Credibility: Ask for the FDA registration number and verify it in the public database. An FDA-registered device has a verifiable public record. An unregistered device carries immense liability that transfers to the operator the moment a client has an adverse event.
-
Irradiance Realities: Ask for measured irradiance at actual treatment distance in mW/cm², and check whether that number comes from independent testing. The OvationULT delivers 65 mW/cm² measured right from the surface. Vendors who quote peak irradiance at contact distance rather than at actual treatment distance are inflating their figures to make up for a lack of design efficiency.
-
Warranty and Service: A device down during peak hours is lost revenue. Ask what the warranty covers, the average service response time, and whether parts are stocked domestically. Body Balance System offers a 5-year warranty on parts and labor for the OvationULT, which is one of the longest coverage periods in the commercial segment.
-
Compliance Boundaries: If a vendor claims recovery time reduction, performance enhancement, or inflammation reduction as direct device claims, they are operating outside their registered scope. Clinical language must be attributed to peer-reviewed research, not the specific device.
FAQ
Q: Can red light therapy be included in a recovery center membership without additional liability concerns?
Yes, provided you use an FDA-registered device and stay within indicated uses: topical heating, temporary relief of minor muscle and joint pain/stiffness, temporary relief of minor arthritis pain, relaxation of muscle spasms, and temporary increase of local circulation.
Q: How many sessions per day can one OvationULT unit realistically handle in a recovery center setting?
At a standard 15-minute session within the 10 to 20 minute range, the unit supports 2 clients per hour. A 12-hour operating day yields approximately 24 sessions at full utilization. Most operators plan for 60 to 75 percent utilization as a conservative revenue baseline, which puts realistic daily sessions at 14 to 18 per unit.
Q: Does adding red light therapy require additional staff in a recovery center?
No dedicated practitioner is required. Sessions run 10 to 20 minutes, the protocol is set by the operator in advance, and clients follow a brief orientation. Front-desk staff can manage RLT rotations alongside standard check-in duties without additional headcount.
Q: How does the OvationULT compare to consumer-grade red light panels in a commercial setting?
The OvationULT is built for high-volume commercial duty, whereas consumer panels often lack FDA registration for commercial applications, commercial-grade duty cycles, business warranty coverage, and verified high irradiance (65 mW/cm²) at actual treatment distance.
Q: What is the best placement of RLT in a recovery center session sequence?
Most operators sequence RLT after training or exertion and before a cold plunge or cryotherapy. The warming and local circulation effects of a 10 to 20 minute session serve as an excellent bridge between the exertion phase and cold contrast therapy.
Sources
-
Global Wellness Institute, "Global Wellness Economy Monitor," 2022 and 2023 editions. https://globalwellnessinstitute.org/industry-research/
-
ISPA (International Spa Association), "ISPA U.S. Spa Industry Study," 2023. https://experienceispa.com/resources/research-tools
-
PubMed / NCBI, photobiomodulation and low-level laser therapy research index. https://pubmed.ncbi.nlm.nih.gov/?term=photobiomodulation+muscle+recovery
-
FDA Device Registration and Listing Database. https://www.fda.gov/medical-devices/how-study-and-market-your-device/device-registration-and-listing
Related Resources
-
How commercial red light therapy works for chiropractic and physical therapy operators
-
Red light therapy in hospitality: what luxury resort operators evaluate
-
Medspa ROI guide: calculating payback on red light therapy hardware
-
Irradiance specs for the OvationULT: what 65 mW/cm2 means for your protocol
-
What "FDA registered" means for commercial red light therapy devices
-
-
 May, 2026NRTL Certification for Red Light Therapy: What It Is, Why It Matters, and How to Verify
May, 2026NRTL Certification for Red Light Therapy: What It Is, Why It Matters, and How to VerifyOSHA requires that electrical equipment used in U.S. workplaces be tested and certified by a Nationally Recognized Testing Laboratory (NRTL), per 29 CFR 1910.303. Red light therapy equipment installed in a commercial setting qualifies as workplace electrical equipment. Yet the certification question rarely appears in vendor conversations or online comparisons. This guide explains what NRTL certification is, which labs are recognized, how it differs from FDA registration, what relevant safety standards apply to light-emitting equipment, how to verify any manufacturer's listing, and what exposure an operator takes on when they skip this due diligence.
NRTL Certification for Red Light Therapy: What It Is, Why It Matters, and How to Verify
Most buyers of commercial red light therapy equipment spend hours comparing wavelength specs and irradiance numbers. Very few ask the question that OSHA requires employers to be able to answer: has this electrical equipment been tested and certified by a Nationally Recognized Testing Laboratory?
For equipment installed in a medspa, chiropractic office, gym, or hotel spa, that question has real consequences tied to workplace safety law, insurance coverage, and liability exposure.
What Is NRTL Certification and Why Does OSHA Require It?
OSHA established the Nationally Recognized Testing Laboratory (NRTL) program under 29 CFR 1910.7 to create a formal pathway for private-sector testing organizations to evaluate products against consensus safety standards. Under 29 CFR 1910.303, OSHA requires that many kinds of electrical equipment be tested and certified by an NRTL before use in the workplace. The requirement applies to virtually every private-sector employer in the United States.
OSHA's NRTL FAQ states the standard: a product must be "accepted, or certified, or listed, or labeled, or otherwise determined to be safe by a nationally recognized testing laboratory recognized pursuant to 1910.7." Without that mark, the employer has no third-party verification and is subject to citation and penalties.
For any commercial facility, a red light therapy bed is a large piece of powered electrical equipment that draws current, generates heat, and runs for extended periods. That profile sits squarely within the product categories OSHA's certification requirement addresses.
Which NRTLs Are Currently Recognized?
OSHA maintains the definitive list of recognized NRTLs. As of the date of this post, the current OSHA NRTL list includes 21 organizations. The most commonly encountered in the context of commercial equipment:
The Recognized NRTLs: Who They Are and What Their Marks Look Like
NRTL
Mark / Abbreviation
Website
UL LLC
UL Listed (circle-UL mark)
ul.com
Intertek Testing Services NA
ETL Listed mark
intertek.com
CSA Group Testing and Certification Inc.
CSA mark / cCSAus
csagroup.org
TUV Rheinland of North America
TUV Rheinland mark
tuv.com
TUV SUD America Inc.
TUV SUD mark
tuvsud.com
Bureau Veritas Consumer Products Services (BVCPS)
BV mark
bureauveritas.com
NSF International
NSF mark
nsf.org
What Is the Difference Between UL Listed and UL Recognized?
UL Listed applies to complete, finished products tested for safe use in their specific product category and end-use environment. The familiar circle-UL label on the finished unit satisfies OSHA's workplace requirement for most electrical equipment categories.
UL Recognized applies to components and subassemblies incorporated into a larger product. A power supply or internal circuit board might carry UL Recognized status, identified by the backwards "RU" mark. A finished product built from UL Recognized components still requires its own full listing to be OSHA-compliant.
A vendor claiming "UL Recognized components" is describing internal parts. When evaluating commercial RLT equipment, ask whether the finished unit carries a Listed certification, not whether its components do.
How Does NRTL Certification Differ from FDA Registration?
These two credentials address entirely different regulatory concerns. Treating them as interchangeable is a mistake that circulates widely in the commercial RLT market.
Two Different Credentials, Two Different Regulators
Credential
Regulating Body
What It Covers
What It Proves
FDA Registration
U.S. Food and Drug Administration
Manufacturing facility and medical device product listing; governed by 21 CFR Part 807
The manufacturer is registered with the FDA and the device is listed; it does NOT mean the device is cleared or approved for any specific therapeutic claim
NRTL Certification (e.g., UL Listed, ETL Listed)
OSHA (via recognized testing labs)
Electrical safety of the finished product against relevant consensus standards
The product was independently tested and found to comply with product safety standards for safe use in the workplace
FDA 510(k) Clearance
FDA
Substantial equivalence of a device to a legally marketed predicate; required for Class II devices making specific therapeutic claims
The device is cleared to market with specified indications for use
FDA Premarket Approval (PMA)
FDA
Clinical evidence for high-risk Class III devices
The highest level of FDA market authorization
What Safety Standards Apply to Red Light Therapy Equipment?
IEC 60601-1 is the international standard for medical electrical equipment, covering general safety and essential performance requirements including electrical insulation, temperature limits, and mechanical hazards. The current version is IEC 60601-1:2005/AMD2:2020.
IEC 62471 is the standard for photobiological safety of lamps and lamp systems. It classifies light sources into risk groups based on emission profile. Red and near-infrared wavelengths used in RLT equipment typically fall into the Exempt or Risk Group 1 (Low-Risk) categories under IEC 62471 in normal use conditions.
The distinction that matters for buyers: "We follow IEC standards" is a self-declaration. "We hold a UL Listed certification to IEC 60601-1" is a verifiable finding from an independent lab. Ask for the latter.
What Is the Risk for an Operator Who Installs Non-NRTL Equipment?
OSHA Citations. Non-NRTL electrical equipment in a workplace creates a basis for citation under 29 CFR 1910.303. OSHA penalties are tiered by violation severity, from other-than-serious through willful. An inspection triggered by a complaint or routine audit will surface missing certification.
Insurance Claim Denial. Commercial liability and property policies require that installed equipment meet applicable safety standards. If a non-NRTL unit is involved in an incident, the insurer has grounds to deny or reduce the claim. Non-compliant equipment is not treated the same as properly certified equipment at claims time.
Liability Exposure. If someone is injured and the investigation finds the equipment lacked required certification, that gap becomes part of the record. An operator cannot make a reasonable-care argument when a basic, verifiable standard was not met.
For an operator who has invested $59,997 in commercial RLT equipment, verifying certification before purchase costs nothing. Skipping it can cost substantially more.
How Do You Verify a Manufacturer's NRTL Listing?
Step 1: Read the nameplate. Every certified product must display the NRTL's registered mark, the applicable standard, and the certification file number. A missing or illegible mark, or a mark from a lab not on the OSHA NRTL list, is a red flag.
Step 2: Search the NRTL's database. Each major lab maintains a public directory.
NRTL
Verification Resource
URL
UL LLC
UL Product iQ
productiq.ul.com
Intertek / ETL
ETL Listed Mark Directory
CSA Group
CSA Certified Product Listing
csagroup.org/testing-certification/product-listing/
Step 3: Request the listing certificate. A manufacturer with a current NRTL listing can produce the certificate on request. It will name the lab, the product, the applicable standard, and the certification scope. A certificate for a different model in the same product family does not cover the unit you are buying.
Due Diligence Checklist for Commercial RLT Equipment Buyers
Before signing a purchase agreement for any commercial RLT unit, confirm:
-
[ ] Does the finished unit carry a listing mark from an OSHA-recognized NRTL?
-
[ ] Which NRTL issued the certification? Is that lab on the current OSHA NRTL list?
-
[ ] What standards was it tested to (IEC 60601-1, IEC 62471)?
-
[ ] Can the manufacturer produce the listing certificate for the specific model?
-
[ ] Does the physical nameplate display the NRTL mark?
-
[ ] Is the manufacturer FDA registered, and do they disclose their registration number?
-
[ ] Has your facility's electrical configuration been reviewed by a licensed electrician?
Properties like Four Seasons, Fairmont, Bellagio, Aria, and Canyon Ranch run their equipment through in-house engineering review before installation. An independent operator should apply the same standard.
How Does BBS Approach the Certification Conversation?
Body Balance System has been deploying commercial red light therapy equipment in professional settings for over 13 years. The install history includes properties like Four Seasons, Fairmont, Bellagio, Aria, and Canyon Ranch. Luxury hotels and resort spas have in-house engineering teams that evaluate equipment before it enters a facility. Those teams ask certification questions before a purchase order is signed.
BBS is an FDA-registered manufacturer. Registration number 3010627475 is verifiable in the FDA's CDRH database. The OvationULT is listed as an Infrared Lamp for Heating (ILY product code), Class II medical device. Read our full overview on what FDA registered actually means to learn how to verify any manufacturer's status.
On the NRTL question: BBS encourages every buyer to ask their vendor directly, in writing, which NRTL certified the finished product and under which standard, and to request the actual certificate. Ask BBS the same question. A manufacturer that cannot answer clearly is giving you information you need before you commit. The credential should be verifiable, the testing should cover the finished product, and the buyer should never have to take the vendor's word for it.
Frequently Asked Questions
What is an NRTL and who recognizes them?
A Nationally Recognized Testing Laboratory (NRTL) is a private organization OSHA has recognized under 29 CFR 1910.7 as qualified to test and certify products against consensus safety standards. OSHA currently recognizes 21 NRTLs. The complete list is at osha.gov. OSHA recognition is not an endorsement of certified products; it confirms the lab has the qualifications to perform the testing.
Does commercial red light therapy equipment require NRTL certification?
Under 29 CFR 1910.303, electrical equipment used in U.S. workplaces must be certified by a recognized NRTL. A commercial RLT bed installed in a medspa, gym, hotel, or medical office is workplace electrical equipment subject to that requirement. Beyond OSHA, commercial liability insurers typically require that installed equipment meet applicable safety standards.
What is the difference between UL Listed and UL Recognized?
UL Listed covers complete, finished products tested for their specific end-use environment. UL Recognized covers components or subassemblies incorporated into a larger system. A finished product built from UL Recognized components still requires its own UL Listed certification. When evaluating commercial RLT equipment, confirm that the finished unit carries a Listed certification, not just its internal components.
Does FDA registration cover electrical safety?
No. FDA registration under 21 CFR Part 807 addresses the manufacturing facility and device listing in the CDRH database. It does not cover electrical safety or OSHA workplace requirements. NRTL certification and FDA registration address separate regulatory concerns. Neither substitutes for the other.
How do I verify NRTL certification for a specific product?
Check the nameplate on the physical unit for an NRTL mark. Then search the NRTL's public database: UL at productiq.ul.com, ETL at intertek.com/directories/etl-listed-mark/, CSA at csagroup.org/testing-certification/product-listing/. Finally, request the actual listing certificate for the specific model from the manufacturer.
What standards apply to red light therapy equipment specifically?
IEC 60601-1 covers general safety for medical electrical equipment, including insulation, temperature limits, and mechanical requirements. IEC 62471 covers photobiological safety of lamps and lamp systems. Red and near-infrared wavelengths used in RLT typically fall into the Exempt or Risk Group 1 (Low-Risk) categories under IEC 62471.
What are the consequences of installing non-NRTL equipment?
OSHA citations and monetary penalties based on violation classification (serious, willful, or repeated). Potential denial of commercial insurance claims on the basis that the equipment did not meet applicable safety standards. Personal liability exposure if a client or employee is injured and non-certification becomes part of the legal record.
Related Resources
For more on the related compliance landscape for commercial red light therapy, explore our resource library:
-
What Does FDA Registered Actually Mean for a Red Light Therapy Device?
-
Irradiance Due Diligence: How to Read and Verify RLT Manufacturer Claims
-
Photobiomodulation Mechanism: What the Peer-Reviewed Evidence Actually Shows
-
How to Add Red Light Therapy to a Medspa: A Commercial Implementation Guide
-
Reading Peer-Reviewed Research on Red Light Therapy: A Practitioner's Guide
External Sources
-
OSHA NRTL Program, Current List of NRTLs: https://www.osha.gov/nationally-recognized-testing-laboratory-program/current-list-of-nrtls
-
OSHA NRTL FAQ, 29 CFR 1910.7 and 1910.303: https://www.osha.gov/nationally-recognized-testing-laboratory-program/frequently-asked-questions
-
FDA Device Registration and Listing, 21 CFR Part 807: https://www.fda.gov/medical-devices/how-study-and-market-your-device/device-registration-and-listing
-
IEC 62471:2006, Photobiological safety of lamps and lamp systems: https://webstore.iec.ch/en/publication/7076
-
Intertek ETL Listed Mark Directory: https://www.intertek.com/directories/etl-listed-mark/
-
UL Product iQ Certification Database: https://productiq.ul.com
-
CSA Group Certified Product Listing: https://www.csagroup.org/testing-certification/product-listing/
-
-
 May, 2026The State of Commercial Red Light Therapy in 2026: Market Trends Operators Need to Watch
May, 2026The State of Commercial Red Light Therapy in 2026: Market Trends Operators Need to WatchThe commercial red light therapy market has moved past the early-adopter phase. According to Grand View Research, the global market reached $533.8 million in 2025 and is projected to hit $587.5 million in 2026, with a compound annual growth rate of 9.8% through 2033. The B2B segment, covering medspas, wellness centers, hospitality facilities, and clinical practices, accounted for the largest revenue share in 2025. Six forces are reshaping the market right now: accelerating vertical adoption across new facility types, a more complex FDA regulatory landscape, private equity consolidation in adjacent wellness equipment categories, growing pressure for irradiance and methodology transparency, supply chain sourcing risk from white-label proliferation, and a tightening 12-to-24-month window for operators to establish market position before competition intensifies.
How Big Is the Commercial RLT Market in 2026, and How Fast Is It Growing?
The numbers tell a consistent story, even if different research firms measure the market at different scales. Grand View Research places the global red light therapy market at $533.8 million for 2025, growing to $587.5 million in 2026 at a 9.8% CAGR through 2033. The broader red light therapy beds segment shows steeper growth projections: Coherent Market Insights estimates the global beds market at $9.29 billion in 2026, reaching $19.56 billion by 2033 at a 13.2% CAGR. The bed-focused figure reflects a wider product scope that includes consumer formats, but both data sets point in the same direction.
B2B channels, covering medspas, wellness centers, rehabilitation centers, and hospitality facilities, still account for the largest revenue share in absolute terms. The B2C segment (home-use panels and portable devices) now carries the fastest CAGR in the forecast period, per Grand View Research, which reflects the consumer awareness wave feeding commercial demand downstream. North America holds a 44.6% global market share, while Asia Pacific is the fastest-growing region.
The broader wellness economy provides important context. According to the Global Wellness Institute's 2025 Wellness Economy Monitor, the global wellness economy reached $6.8 trillion in 2024 (up 7.9% from 2023), with a projected expansion to $9.8 trillion by 2029. The GWI specifically names infrared light therapy and photobiomodulation among the longevity and biohacking approaches now ubiquitous in fitness centers, spas, and resorts. RLT is not a niche add-on. It is becoming a standard facility offering.
Commercial RLT Market Size and Growth Projections 2025-2033
Metric
Value
Source
Global RLT market, 2025
$533.8 million
Grand View Research
Global RLT market, 2026 (projected)
$587.5 million
Grand View Research
Global RLT market, 2033 (projected)
$1,133.1 million
Grand View Research
CAGR 2026-2033
9.8%
Grand View Research
RLT beds segment, 2026 (projected)
$9.29 billion
Coherent Market Insights
RLT beds segment, 2033 (projected)
$19.56 billion
Coherent Market Insights
RLT beds CAGR 2026-2033
13.2%
Coherent Market Insights
North America market share, 2025
44.6%
Grand View Research
Global wellness economy, 2024
$6.8 trillion
Global Wellness Institute
What Verticals Are Driving Commercial RLT Adoption?
Medspas are the anchor vertical. Consumer demand for non-surgical, non-injectable service options has grown at mid-to-high single-digit rates, according to data reviewed by the American Med Spa Association. Red light therapy slots in as a standalone service, a membership anchor, or an add-on to aesthetic treatments. The Fortune Business Insights light therapy market report notes that dermatology clinics and medical spas are primary buyers of commercial red light therapy devices, with red light holding approximately 34% of the total light therapy market share.
Hospitality is the fastest-growing commercial segment from a smaller base. Hotels and hospitality facilities are explicitly part of the B2B segment driving growth, per Grand View Research. The Global Wellness Institute notes that governments and hospitality brands are investing in therapeutic modalities to differentiate properties as wellness tourism expands. Properties such as Four Seasons, Fairmont, and Canyon Ranch have deployed commercial-grade red light therapy beds as premium amenity offerings, recognizing that equipment quality reflects on the property's overall wellness positioning.
Chiropractic, rehabilitation, and recovery-focused fitness facilities represent the next adoption wave. The Fortune Business Insights report cites rising demand from orthopedic and chiropractic centers as a specific growth driver. For these operators, the business case is direct: sessions run 10-20 minutes, setup is minimal, and the modality integrates into existing scheduling without adding staff. The Grand View Research data notes Precor partnered with Wellness USA in October 2024 to distribute RLT equipment across fitness facilities, spas, and hotels globally, signaling the mainstream fitness equipment channel treating RLT as a standard offering. Muscle recovery applications are projected to show the fastest CAGR among application segments through 2033.
Vertical Adoption Curve for Commercial Red Light Therapy 2024-2026
Vertical
Adoption Stage (2026)
Primary Use Case
Key Growth Driver
Medspas
Mature / mainstream
Non-invasive wellness, recovery add-on
Non-surgical service demand
Luxury hospitality
Early growth
Premium amenity, spa differentiation
Wellness tourism expansion
Chiropractic practices
Early adoption
Pain management support, recovery
Non-pharmaceutical demand
Rehabilitation centers
Early adoption
Recovery, musculoskeletal support
Clinical PBM evidence base
Performance / recovery gyms
Emerging
Pre- and post-workout recovery
Athletic performance interest
Standard fitness clubs
Pre-adoption
Member retention add-on
Competitive differentiation
How Is the FDA Regulatory Landscape Shifting?
The FDA landscape for commercial red light therapy devices operates on two distinct tracks.
The first is the ILY product code (Infrared Lamp for Heating). Devices listed under ILY are Class II medical devices registered with the FDA. Registration means the manufacturer has submitted facility and device information to the FDA's device database, not that the device has undergone premarket review for specific clinical claims. ILY registration is the appropriate pathway for full-body commercial beds used for topical heating, temporary relief of minor muscle and joint pain and stiffness, temporary relief of minor arthritis pain, relaxation of muscle spasm, and temporary increase of local circulation. Body Balance System holds FDA Registration number 3010627475 under the ILY product code.
The second track is the OLH product code, covering phototherapy devices for skin conditions. OLH devices go through the 510(k) premarket notification pathway and can make specific claims tied to their cleared intended use, such as treatment of facial wrinkles or acne. The FDA's public 510(k) database shows a meaningful uptick in OLH filings, with multiple Shenzhen-based manufacturers receiving clearances in late 2025 and early 2026 (see K253400 and K252603).
For operators: the commercial market now contains devices at two regulatory tiers with different permitted claims frameworks. Neither designation is superior. The compliance requirement is that any device's registration or clearance number should be verifiable in the FDA's public database, and marketing claims should align with the device's regulatory classification. Operators should request the FDA establishment registration number and verify it directly. For more detail, see the BBS resource on what FDA registered actually means for red light therapy devices.
Why Is Consolidation Happening, and What Does It Mean for Buyers?
The medspa sector has entered an acknowledged consolidation cycle. According to data from the American Med Spa Association, private equity-backed platforms and management service organizations remained "highly active" in 2025, particularly in Florida, Texas, and California. Named platforms in active recapitalization include Empower Aesthetics (Shore Capital), Alpha Aesthetics (Thurston Group), and Advanced MedAesthetic Partners (Leon Capital). Greater than 90% of medspas remain independently owned, but PE is building regional scale through add-on acquisitions at the platform level.
When PE groups acquire medspa platforms, they standardize operations, including equipment suppliers. A platform that adopted a particular equipment brand during its independent phase carries that decision into every add-on acquisition. Operators who are potential acquisition targets should recognize that their equipment roster becomes part of the diligence story. Equipment with verifiable regulatory documentation, a demonstrable service track record, and a clear warranty structure is easier to underwrite than equipment from brands with thin operational histories.
Supply chain dynamics add another layer. A growing share of devices entering the market through direct-import channels originate from Chinese factories and are sold through white-label brands. This is not inherently a quality issue; many established brands source manufacturing offshore with strong specifications. The risk is when operators purchase devices with minimal documentation, warranties under 12 months (vs. the commercial standard of five years), and no verifiable FDA registration. For a deeper examination, see our guide on irradiance due diligence for commercial red light therapy buyers.
Where Is the Irradiance and Methodology Conversation Heading?
Irradiance transparency is one of the most active discussions in the commercial RLT channel right now, and it is being driven by a measurement methodology problem.
Broadband solar meters, which are low-cost handheld sensors used widely in the consumer and commercial RLT market, consistently overstate therapeutic irradiance relative to accredited laboratory measurement. Published data from ISO/IEC 17025-accredited photometric testing shows that broadband solar meter readings typically run 2.2 to 2.5 times higher than what the same device delivers when measured at therapeutic red and near-infrared wavelengths by a calibrated laboratory instrument traceable to NIST standards. This means a device advertised at "150 mW/cm²" based on a solar meter reading may deliver closer to 60-68 mW/cm² when measured with accredited lab instruments at therapeutic wavelengths. This is not a regulatory violation; irradiance measurement methodology is not currently standardized by FDA for ILY-class devices. But it creates a material information gap for buyers comparing equipment specifications.
The pressure for standardization is building from multiple directions. A subset of manufacturers has begun publishing ISO/IEC 17025-accredited lab reports for their devices. Peer review publications examining dose-response relationships in photobiomodulation are increasingly explicit about the need for standardized measurement protocols. NRTL (Nationally Recognized Testing Laboratory) certification bodies, including UL and ETL, provide electrical safety listings for commercial equipment but do not currently certify irradiance output. Some manufacturers voluntarily pursue NRTL listings as a signal of overall quality commitment. For operators who want to understand what the irradiance numbers in equipment specifications actually mean, our explainer on peer-reviewed research and commercial RLT standards provides context on how published PBM research actually characterizes dose.
For commercial operators, the practical implication is straightforward: ask any prospective equipment supplier how their irradiance figure was measured, at what distance, and whether the data comes from an accredited laboratory or a broadband solar meter. Contact irradiance at the point of client contact matters most. Body Balance System, for example, publishes 65 mW/cm² at contact for the OvationULT. The methodology behind any irradiance claim should be part of the procurement conversation for any commercial buyer. For guidance on what to ask, see how to add red light therapy to a medspa.
The broader photobiomodulation research community has also continued to accumulate evidence. Published research on the mechanism of action, specifically cytochrome c oxidase activation and ATP production in mitochondria, is well-established in the academic literature. For a primer on the cellular mechanism, see our resource on the photobiomodulation mechanism and how red light therapy works.
What Does the Next 12-24 Months Look Like for Commercial Operators?
Several forces are converging to make the next two years a consequential window for commercial operators considering red light therapy equipment.
On the demand side, the signals are consistent. According to the ISPA Foundation's 2025 Consumer Snapshot Study (PricewaterhouseCoopers, 1,000 U.S. respondents), 85% of spa-goers view visits as self-care, with younger consumers showing stronger acceptance of technology-assisted services. The North American spa industry generated $22.5 billion in revenue in 2024, per ISPA's 2025 U.S. Spa Industry Study as reported by Spas of America, its third consecutive year of expansion. Infrared and light therapy are specifically listed among longevity and biohacking-inspired services driving that growth.
On the supply side, the commercial equipment channel is becoming more crowded. Growing consumer demand and a relatively low barrier to importing and rebranding devices means new brands enter the market steadily. Many are consumer-grade products repositioned for commercial sale. For operators, the relevant questions go beyond specifications: Does the manufacturer have a commercial installation track record? Is the equipment backed by a multi-year commercial warranty? Does the manufacturer carry verifiable FDA registration?
The PE rollup activity in medspas reinforces this point. Equipment decisions are increasingly made at the platform level, not just the individual location. Operators building toward a multi-site model or positioning for a future sale should weigh equipment documentation, warranty structure, and manufacturer longevity alongside raw specifications. BBS, for example, has supported commercial installations for 13 years across properties including Bellagio, Aria, Four Seasons, Fairmont, and Canyon Ranch, and backs every OvationULT with a 5-year white-glove warranty.
Frequently Asked Questions
What is the size of the commercial red light therapy market in 2026?
According to Grand View Research, the global red light therapy market is projected to reach $587.5 million in 2026, up from $533.8 million in 2025. The broader red light therapy beds segment is estimated at $9.29 billion in 2026 by Coherent Market Insights, reflecting a wider product scope that includes all commercial and consumer bed formats globally.
What is the difference between FDA registration and FDA 510(k) premarket notification for red light therapy devices?
FDA registration means the manufacturer has submitted facility and device listing information to the FDA's establishment registration database. It does not involve premarket review of clinical claims. The 510(k) premarket notification pathway requires the manufacturer to demonstrate substantial equivalence to a predicate device and, if granted, permits specific clinical claims tied to the cleared intended use. The two designations serve different regulatory purposes and permit different marketing claims. For a detailed breakdown, see our guide on what FDA registered actually means.
Which verticals are growing fastest in commercial RLT adoption?
Medspas are the largest current adopter vertical. Hospitality (luxury hotels and resort spas) and performance-focused recovery facilities are showing the fastest growth rates from a smaller base. Chiropractic and rehabilitation practices represent a third wave of adoption, driven by growing patient demand for non-pharmaceutical modalities and an expanding base of published clinical evidence on photobiomodulation.
How should operators evaluate irradiance specifications when comparing commercial equipment?
Ask the manufacturer whether their irradiance figure was measured with a broadband solar meter or with a calibrated, accredited laboratory instrument (ISO/IEC 17025). Broadband solar meters consistently overstate therapeutic irradiance relative to accredited lab measurements by a factor of 2.2 to 2.5. Request documentation of the measurement distance and methodology. Contact irradiance at the point of client use is the most operationally relevant figure. View our irradiance due diligence guide for a structured evaluation framework.
What does PE consolidation in medspas mean for equipment purchasing decisions?
As private equity-backed platforms acquire multi-location medspa operators, equipment decisions that were previously made at the individual site level are increasingly made at the platform level. Operators positioning for acquisition or multi-site growth benefit from choosing equipment with verifiable regulatory documentation, commercial warranty coverage, and a manufacturer with a demonstrable multi-year service track record. These factors reduce diligence friction in a transaction process.
How long is a typical commercial red light therapy session?
Commercial red light therapy sessions typically run 10-20 minutes. This range allows operators to schedule approximately two clients per hour per unit, net of transition time, at a 15-minute session. Session length within the 10-20-minute range should be set based on the operator's protocol and the specific device's irradiance output. For specific revenue models, see our commercial red light therapy pricing guide.
Is the RLT market likely to face increased regulatory scrutiny?
The current trajectory suggests continued regulatory attention on claims standards more than on device registration requirements. The FDA has issued 510(k) clearances for an increasing number of OLH-code devices with specific cosmetic and dermatological claims. The ILY-code registration pathway remains the appropriate route for full-body commercial beds without 510(k) clearance. Operators and manufacturers who stay within their device's permitted claims scope are best positioned as oversight evolves.
Citations
-
Grand View Research. "Red Light Therapy Market Size, Share and Industry Report, 2033." https://www.grandviewresearch.com/industry-analysis/red-light-therapy-market-report
-
Coherent Market Insights. "Red Light Therapy Beds Market Size and Forecast, 2026-2033." https://www.coherentmarketinsights.com/industry-reports/red-light-therapy-beds-market
-
Global Wellness Institute. "The Global Wellness Economy Hits a Record $6.8 Trillion and Is Forecast to Reach $9.8 Trillion by 2029." November 19, 2025. https://globalwellnessinstitute.org/press-room/press-releases/the-global-wellness-economy-hits-a-record-6-8-trillion-and-is-forecast-to-reach-9-8-trillion-by-2029/
-
ISPA Foundation / International SPA Association. "Self-Care, Stress Relief and Tech Appeal: ISPA's 2025 Consumer Snapshot Study." June 26, 2025. https://experienceispa.com/press-releases/self-care-stress-relief-and-tech-appeal-ispas-2025-consumer-study-reveals-what-drives-spa-visits/
-
American Med Spa Association / Xite. "Med Spa M&A and Private Sales: A Look Back at 2025 and What Lies Ahead." May 15, 2026. https://www.americanmedspa.org/news/med-spa-ma-and-private-sales-a-look-back-at-2025-and-what-lies-ahead/
-
Fortune Business Insights. "Light Therapy Market Size, Industry Share, Forecast 2034." https://www.fortunebusinessinsights.com/light-therapy-market-106935
-
Join the List
- Choosing a selection results in a full page refresh.
- Opens in a new window.


























